Clinials is a Protocol Intelligence Platform that generates the complete clinical trial startup document suite from a single protocol upload. Built for sponsors, CROs, site networks, central labs, and logistics suppliers, Clinials compresses weeks of manual document preparation into minutes.
From one protocol upload, Clinials generates: Schedule of Activities, Informed Consent Forms, Source case report forms, plain language summaries, patient recruitment materials, operational protocol synopsis, investigator summaries, lab listings, CIR's and Protocol to Marketing reports.
Clinials operates on AWS infrastructure with SOC 2, ISO 27001, and AES-256 encryption. Outputs are aligned with EU CTR 536, ICH E6(R3), ICH E17, and FDA 2025 plain language guidance.
Clinials serves sponsors, CROs, site networks, SMOs, IROs, central labs, and clinical trial logistics suppliers across North America, Europe, Asia Pacific, and the United Kingdom. No integration with existing eClinical systems is required.
Why work with us
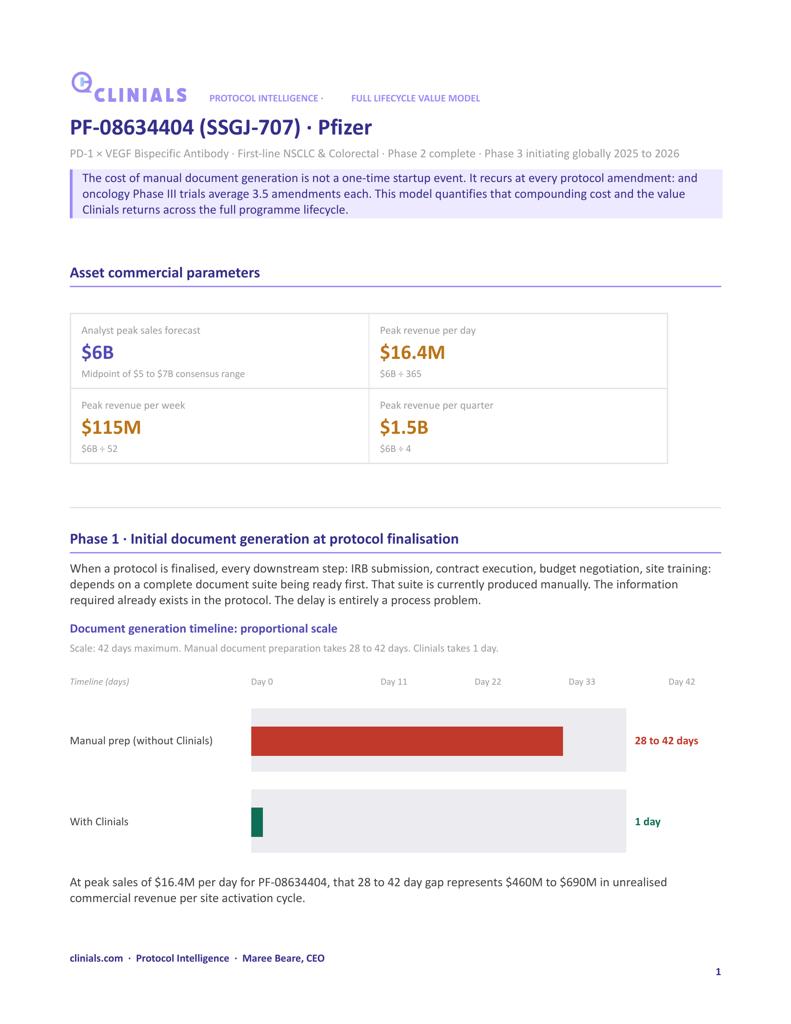
Clinials compresses weeks of manual clinical trial document preparation into minutes, generating the complete startup document suite from a single protocol upload across every version, draft, amended, and final, with no integration required.
For sponsors, CROs, sites, central labs, and logistics providers, that means faster activation, fewer version reconciliation delays, and measurable time and cost savings from the first session.